Welcome to the unit on Thermochemical principles and atomic structure and theory. This unit is assessed externally through AS91391 (AS 3.4) and is worth 5 credits.
|
Unit Outline: A copy of the relevant achievement standard is available on the right here.
For an interpretation of the requirements of the standard in an outline form please download the outline file. |
| ||||||||||||
|
Electron Configuration:
You must be able to write electron configurations (using s,p,d notation) for atoms or ions of the first 36 elements. "Electron configuration of atoms and ions of the first 36 elements (using s,p,d notation)." BestChoice: AS3.4 - Particle Properties: |
Other Files:
|
| ||||||
|
Periodic Trends:
You must be able to define, discuss trends and compare values for electronegativity, ionisation energies, atomic radii, and ionic radii. "Periodic trends in atomic radius, ionisation energy, and electronegativity, and comparison of atomic and ionic radii." BestChoice: AS3.4 - Particle Properties: |
Download Links:
| |||||||||||||
|
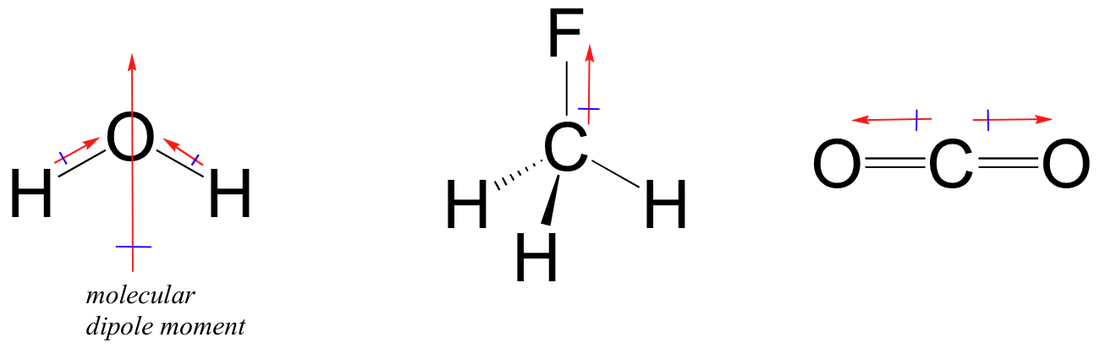
Shape, Polarity and Lewis Structures:
You must be able to draw Lewis diagrams for structures; including knowing common exceptions to the octet rule. You should then be able to use this structure along with the VSEPR theory to predict or explain molecules shape and polarity. "Lewis structures and shapes (up to six electron pairs about the central atom for molecules and polyatomic ions, including those with multiple bonds), polarity of molecules." BestChoice: AS3.4 - Particle Properties: |
Other Files:
Useful overview notes on shapes and on answering "explain" type questions (Source: www.nobraintoosmall.co.nz) |
Download Links:
| ||||||||||||
|
Attractive Forces between Atoms, Ions, and Molecules
You must be able to link the bonding of substances with their properties. You should be able to differentiate between types of solids and types of intermolecular bonds. You will also need to be able to explain solubility trends (including giving descriptions of what happens on the molecular level) and make predictions based on bond types. "Attractive forces between atoms, ions, and molecules. These will include ionic bonds, covalent bonds, and intermolecular attractions due to temporary dipoles and permanent dipoles (including hydrogen bonding)." BestChoice: AS3.4 - Particle Properties: |
Other Files:
|
Download Links:
|